Recent Blog
-

Comprehensive Guide on How to Manage Stress Understanding Causes Symptoms Precautions and Treatments
-

Harmonizing Health: Navigating Time, Lifestyle, and Anxiety
-

Embracing Rainy Season Wellness: Cooper Pharma's Commitment to Health and Rainy Day Delights
-
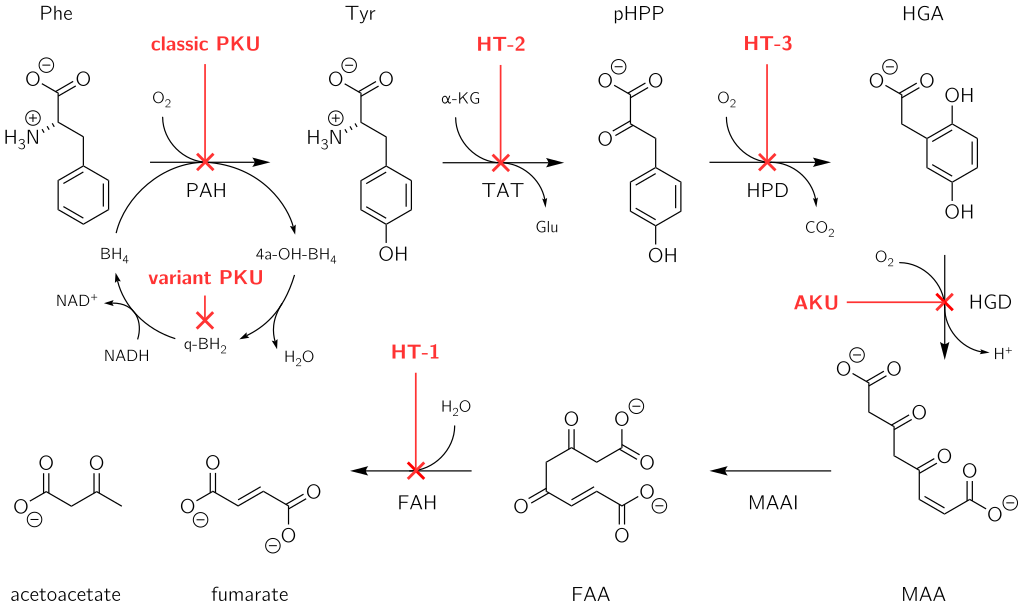
Alkaptonuria: The Rare Inherited Disorder Revealed
-

The Restorative Power of Sleep: Nurturing Overall Wellness
-

Sweet Liberation: Unleashing the Power of Reduced Sugar for Gym Enthusiasts
-

Unleashing the Power of Innovation: Cooper Pharma's Journey to Excellence in the Indian Pharmaceutical Industry
-

Cooper Pharma: Inspiring Health and Wellness through Pharmaceutical Excellence
-

Embrace Life: Uniting for a Tobacco-Free Future on World No Tobacco Day
-

The Powerhouse Unveiled: A Comprehensive Look at India's Pharmaceutical Industry and Its Diverse Manufacturing Landscape
-

Understanding Hypertension: A Silent Threat on World Hypertension Day
-

Achieving Optimal Health and Wellness: 7 Simple Tips to Follow
-

World Lupus Day: Understanding, Caring, and Supporting Those Affected by Lupus
-

Combating Malaria: Progress and Challenges on World Malaria Day 2023
-

Exporting High-Quality Medicines Worldwide: Cooper Pharma's Impact on the Global Pharmaceutical Market
-

An Overview of India's Pharmaceutical Industry and Cooper Pharma's Contribution to It
-

Affordable and Accessible Healthcare Solutions for All
-

Prioritizing Health and Wellbeing on World Health Day and Beyond
-
.png)
What is PharmTech?
-

International Day of the Unborn Child: Cooper Pharma's Commitment to Promoting Prenatal Health and Well-Being
-

Tuberculosis: Symptoms, Diagnosis, and Treatment
-

Understanding Down Syndrome and Cooper Pharma's Efforts to Support Individuals with the Condition

Date:-
